
Atoms can be transformed into different elements in nuclear reactions, which change an atom's atomic number.Īlmost all of the baryonic matter of the universe is composed of chemical elements (among rare exceptions are neutron stars). Two or more atoms of the same element can combine to form molecules, in contrast to chemical compounds or mixtures, which contain atoms of different elements. For example, oxygen has an atomic number of 8, meaning that each oxygen atom has 8 protons in its nucleus.

Chemical elements are identified by the number of protons in the nuclei of their atoms, known as the element's atomic number. The basic particle that constitutes a chemical element is the atom. The ready-to-use Exit Slip reproducible for this video can be downloaded in and formats.A chemical element is a chemical substance that cannot be broken down into other substances by chemical reactions. Teachers could collect and review the answers provided to assess the depth of student understanding on this topic.

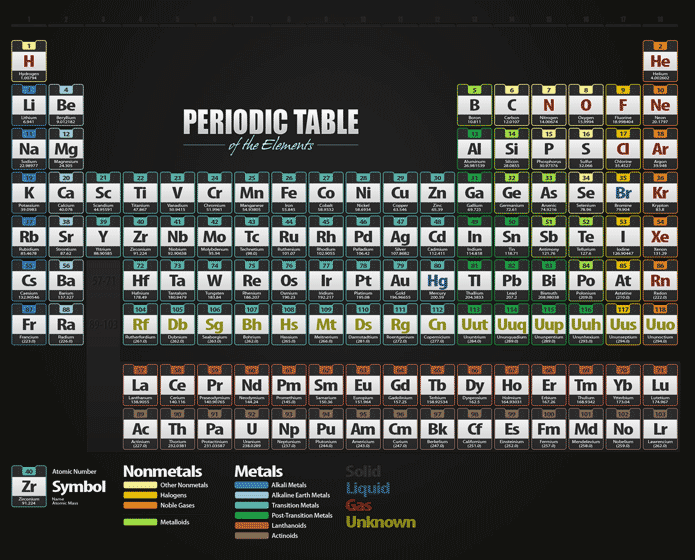
Teachers could have students engage in a think-pair-share to address such questions as “what are elements?,” “how are elements discovered?,” “how do elements get their names?,” “what is the most important element?,” “what is the most valuable element?,” etc. Before watching the video and reading the article, teachers could introduce this topic by engaging students in a discussion to activate their prior knowledge related to elements and the periodic table.Concepts introduced include Periodic Table of Elements, chemical elements, periods, groups, atomic number, metals and nonmetals. This video and article can be used to support teaching and learning of Chemistry and History & World Studies related to the periodic table.Long after his death, Dmitri Mendeleev had an element named after him: Mendelevium, atomic number 101. He even predicted a very rare element called technetium! Where is gallium? That’s right, one step away from aluminum! And it turned out that Mendeleev’s predictions about the properties of this element were spot-on! He also predicted the existence of at least 5 other elements. Sure enough, a few years later, the French chemist Paul-Émile Lecoq de Boisbaudran discovered gallium. But Mendeleev was able to predict many of its properties based on the properties of the elements surrounding it. This place was one step away from aluminum. For example, he set aside a place for eka-aluminum. When Mendeleev devised the periodic table, he left plenty of blank spaces and dashes. But in Mendeleev’s time, scientists only knew of 63 elements. And he was right! Today, the periodic table has 118 elements. Unlike those other scientists, Mendeleev predicted that there would be more chemical elements to come. So why was Mendeleev’s idea the one that lasted? Some of these scientists had even grouped elements with similar properties together. Other scientists had listed the elements. Mendeleev was not the first person to try to organize the elements. What is so special about Mendeleev’s table? Elements on the right-hand side are generally non-metals. Elements on the left-hand side of the periodic table are generally metals. You can also use the table to quickly find an element’s symbol and atomic mass. In the table, elements are arranged based on their atomic number, electron configurations, and chemical properties. Here is an image to help you remember that groups run vertically and periods run horizontally (© 2019 Let’s Talk Science).
